Biology Forum › Microbiology › PPFD and kWh conversion
- AuthorPosts
- March 16, 2017 at 7:33 pm #18375
 agoff3Participant
agoff3ParticipantHi, I am having some problems trying to understand light conversion.
I am trying to work out what 25 umol m-2 s-1 would be in kwh and I am using a cyanobacteria that grows at 470nm.How do I approach this? From what I understand, one unit is measuring brightness and the other energy, and I can’t figure out how to get from one to the other?
If anyone has any resources they could point me to, id be really grateful. Thank you - August 14, 2017 at 3:08 pm #116280
 VmedvilParticipant
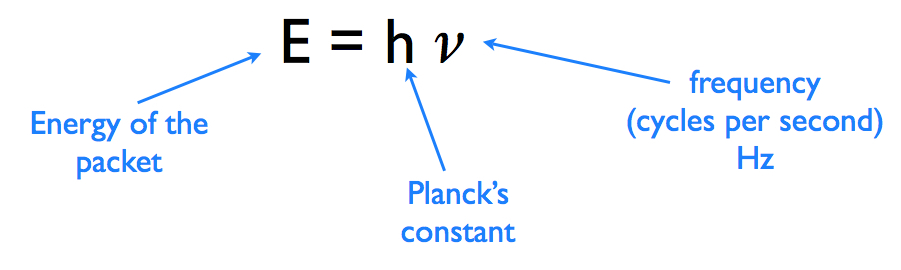
VmedvilParticipantWell, you are going to need to use the physics equation E=h*C/ λ to convert the light wavelength to energy per photon, first convert nano-meters into meters then plug this into the equation modified for wavelength.
E= Energy in (joules per photon)
C = Speed of Light in (meters per second)
h = Plank’s constant
λ = wavelength in meters

Next, Remember that photons hit one atom or Atoms per photon a physics concept called the photoelectric effect. so, take the Energy per photon times how many moles you have or ΣE = E * (Atoms) , Always remember to convert your micro-moles into Atoms.
E = Energy per Photon (Joules per photon)
Atoms = How many atoms are being hit by photons in (Atoms * Photons)
ΣE = Sum of Energy of all photons hitting your atoms in (Joules)
Lastly, you can convert the joules into another unit of energy being kilowatt hours
- AuthorPosts
You must be logged in to reply to this topic.